Volume 1, Issue 3
Kinetic studies of oxidation of Ethambutol in basic media
Author(s): Sandipsingh Gour1, R.P. Phase2, B R Agarwal3, Mazahar Farooqui4*
- Sant Ramdas College, Ghansawangi, Dist Jalna(MS) India
- Lal Bahadur Shastri College, Partur, Dist. Jalna( MS) India
- J E S College, Jalna (MS) India
- Rafiq Zakaria College for Women, Aurangabad( MS) India,
.
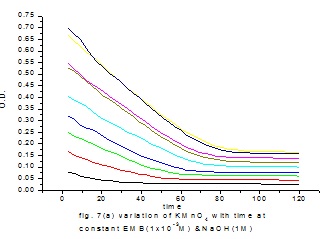
Abstract: The oxidation of pyrazinamide in acidic media is carried out using potassium permanganate as a oxiding agent. The reaction was monitored using UV-Visible spectrophotometer at 525 nm. It was found to be zero order with respect to oxidant,, fractional order with respect to hydrogen ion concentration and first order with respect to substrate. The thermodynamic parameters(were determinied . The average (ΔG#) was found to be 86.508 KJ/mol. The values ΔS# was found to be -0.1354 KJ/mole and energy of activation was found to be 46.7709 KJ/mole. A suitable mechanism is proposed based on the experimental conditions.
Related Graphics: Click here for more graphics
Download Full Article : Click Here
