Volume 1, Issue 4
Polymer Supported Sodium Chromate Oxidation of 1-Phenylethanol: A Kinetic Mechanistic Study
Author(s): Vilas Y. Sonawane1*
Department of Chemistry, B.Raghunath Arts, Commerce and Science, College, Parbhani (MS) India 431401
Related Figures:
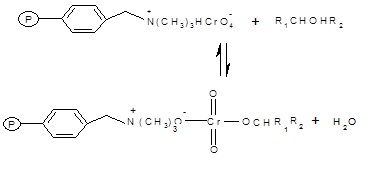 The polymer supported reagent reacts with a molecule of 1-Phenylethanol to form a chromate ester.
The polymer supported reagent reacts with a molecule of 1-Phenylethanol to form a chromate ester.
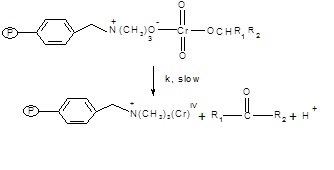 The ester formed will decompose into ketone and the intermediate chromium
(IV) will be formed in the second and slow step.
The ester formed will decompose into ketone and the intermediate chromium
(IV) will be formed in the second and slow step.
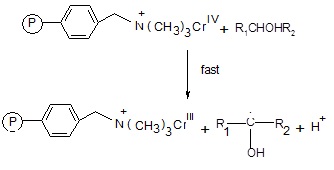 The intermediate chromium (IV) thus reacts with another alcohol molecule to produce a free radical species.
The intermediate chromium (IV) thus reacts with another alcohol molecule to produce a free radical species.
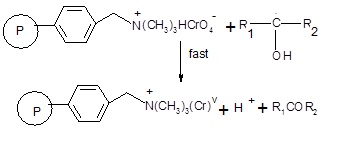 Subsequently the free radical will react with another oxidant site in the
polymeric reagent in a fast step leading to the formation of chromium (V).
Subsequently the free radical will react with another oxidant site in the
polymeric reagent in a fast step leading to the formation of chromium (V).
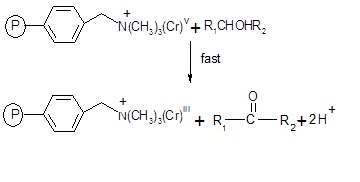 The intermediate chromium (V) in the last step reacts with 1-phenylethanol produce acetophenone.
The intermediate chromium (V) in the last step reacts with 1-phenylethanol produce acetophenone.